Effectiveness and cost-effectiveness of INSPIRatory musclE training (IMT) for reducing postoperative pulmonary complications (PPC): a sham-controlled randomised controlled trial (RCT): INSPIRE
Sponsor: University Hospitals Bristol
Status: In close down, pending publication
After major operations, some patients develop complications of the lungs, including pneumonia. These complications can be serious. They may result in long stays in hospital, may prolong recovery from surgery and may reduce patients’ quality of life. Lung complications are common, affecting on average one in ten patients, with the risk for a particular individual depending on their current health and the type of surgery they are having.
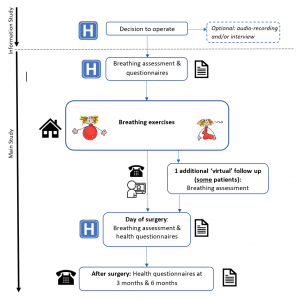
The INSPIRE study is investigating whether a series of daily breathing exercises that aim to improve the strength and endurance of the muscles in the chest can help to reduce the chances of getting a lung complication after surgery.
The aim of the INSPIRE study is to compare exercises performed using a hand-held device held up to the mouth, through which patients breathe in and out, with recommended exercises that are provided in an information leaflet and are used in current care. Training will take approximately 15 minutes twice a day and can be performed at home whilst sitting down. Some studies have shown that training for as little as two weeks before a major operation halves the risk of lung complications.
The INSPIRE study will involve 2500 adult patients undergoing operations in the chest or stomach recruited from several hospitals across the UK. The study design includes an internal pilot phase to ensure deliverability of the full randomised controlled trial (RCT). Patients will be followed up for six months after randomisation and will be asked to complete quality of life questionnaires.
The INSPIRE study is funded by the NIHR HTA Programme (project number 16/140/07). The views expressed are those of the authors and not necessarily those of the NHS, the NIHR HTA or the Department of Health.
PIL diagram new_v2.0_22.07.2020_1
Contact Information
Chief Investigator: Dr Maria Pufulete
Trial Co-Ordinator: Emma Bridgeman
E-mail: inspire-study@bristol.ac.uk